-
September
-
Full breakfast
-
Seamus Heaney
-
Superman
-
2013 Ghouta attacks in Syria
-
Stone paper
-
Look Back in Anger
-
Emmy Award
-
Pun
-
Dolce & Gabbana
-
Russia
-
Stock market bubble
-
Rare earths
-
Sophia Loren
-
Steganography
-
Deindustrialization
-
Subject-auxiliary inversion
-
Phrasal verb
-
Labyrinth
-
Goalkeeper (football)
-
The Decameron
-
Umberto Eco
-
Taser
-
Territorial claims in the Arctic
-
Google Glass
-
Pizza
-
Linux Operating System
-
Augmented reality
-
Charlie Chaplin
-
Lincoln (film)
-
Diwali
|
WIKIMAG n. 10 - Settembre 2013
Rare earths

Text is available under the
Creative Commons Attribution-ShareAlike License; additional
terms may apply. See
Terms of
Use for details.
Wikipedia® is a registered trademark of the
Wikimedia Foundation,
Inc., a non-profit organization.
Traduzione
interattiva on/off
- Togli il segno di spunta per disattivarla

 As
defined by
IUPAC, rare earth elements ("REEs") or rare earth metals
are a set of seventeen
chemical elements in the
periodic table, specifically the fifteen
lanthanides plus
scandium and
yttrium.[2]
Scandium and yttrium are considered rare earth elements since they tend
to occur in the same
ore deposits
as the lanthanides and exhibit similar chemical properties. As
defined by
IUPAC, rare earth elements ("REEs") or rare earth metals
are a set of seventeen
chemical elements in the
periodic table, specifically the fifteen
lanthanides plus
scandium and
yttrium.[2]
Scandium and yttrium are considered rare earth elements since they tend
to occur in the same
ore deposits
as the lanthanides and exhibit similar chemical properties.
Despite their name, rare earth elements (with the exception of the radioactive
promethium) are
relatively plentiful in the
Earth's crust, with
cerium
being the 25th most abundant element at 68 parts per million (similar to
copper).
However, because of their
geochemical properties, rare earth elements are typically dispersed
and not often found concentrated as
rare earth minerals in economically exploitable ore deposits.[3]
It was the very scarcity of these minerals (previously called "earths")
that led to the term "rare earth". The first such mineral discovered was
gadolinite, a
compound of cerium,
yttrium,
iron,
silicon
and other elements. This mineral was extracted from a mine in the
village of
Ytterby
in Sweden;
several of the rare earth elements bear names derived from this
location.
radioactive
promethium) are
relatively plentiful in the
Earth's crust, with
cerium
being the 25th most abundant element at 68 parts per million (similar to
copper).
However, because of their
geochemical properties, rare earth elements are typically dispersed
and not often found concentrated as
rare earth minerals in economically exploitable ore deposits.[3]
It was the very scarcity of these minerals (previously called "earths")
that led to the term "rare earth". The first such mineral discovered was
gadolinite, a
compound of cerium,
yttrium,
iron,
silicon
and other elements. This mineral was extracted from a mine in the
village of
Ytterby
in Sweden;
several of the rare earth elements bear names derived from this
location.
List
A table listing the seventeen rare earth elements, their
atomic number and symbol, the etymology of their names, and their
main usages (see also
Applications of lanthanides) is provided here. Some of the rare
earths are named after the scientists who discovered or elucidated their
elemental properties, and some after their geographical discovery.
| Etymology |
Selected applications |
| 21 |
Sc |
Scandium |
from
Latin
Scandia (Scandinavia),
where the first rare earth ore was discovered. |
Light
aluminium-scandium alloys for aerospace components, additive
in
metal-halide lamps and
mercury-vapor lamps,[4]
radioactive tracing agent in oil refineries |
| 39 |
Y |
Yttrium |
after the village of
Ytterby, Sweden, where the first rare earth ore was
discovered. |
Yttrium aluminium garnet (YAG) laser, yttrium vanadate (YVO4)
as host for europium in TV red phosphor,
YBCO
high-temperature superconductors,
Yttria-stabilized zirconia (YSZ),
yttrium iron garnet (YIG)
microwave filters,[4]
energy-efficient light bulbs,[5]
spark plugs, gas mantles, additive to steel |
| 57 |
La |
Lanthanum |
from the Greek "lanthanein", meaning to be hidden. |
High
refractive index and alkali-resistant glass, flint, hydrogen
storage, battery-electrodes,
camera lenses,
fluid catalytic cracking catalyst for oil refineries |
| 58 |
Ce |
Cerium |
after the dwarf planet
Ceres, named after the Roman goddess of agriculture. |
Chemical
oxidizing agent, polishing powder, yellow colors in glass
and ceramics, catalyst for
self-cleaning ovens,
fluid catalytic cracking catalyst for oil refineries,
ferrocerium flints for lighters |
| 59 |
Pr |
Praseodymium |
from the Greek "prasios", meaning leek-green, and
"didymos", meaning twin. |
Rare-earth magnets,
lasers, core material for
carbon arc lighting, colorant in
glasses and
enamels, additive in
didymium glass used in
welding goggles,[4]
ferrocerium firesteel (flint) products. |
| 60 |
Nd |
Neodymium |
from the Greek "neos", meaning new, and "didymos",
meaning twin. |
Rare-earth magnets,
lasers, violet colors in glass and ceramics,
didymium glass,
ceramic capacitors |
| 61 |
Pm |
Promethium |
after the
Titan
Prometheus, who brought fire to mortals. |
Nuclear batteries |
| 62 |
Sm |
Samarium |
after
Vasili Samarsky-Bykhovets, who discovered the rare earth ore
samarskite. |
Rare-earth magnets,
lasers,
neutron capture,
masers |
| 63 |
Eu |
Europium |
after the continent of
Europe. |
Red and blue
phosphors,
lasers,
mercury-vapor lamps,
fluorescent lamps,
NMR relaxation agent |
| 64 |
Gd |
Gadolinium |
after
Johan Gadolin (1760–1852), to honor his investigation of
rare earths. |
Rare-earth magnets, high refractive index glass or
garnets,
lasers,
X-ray tubes,
computer memories,
neutron capture,
MRI contrast agent,
NMR relaxation agent,
magnetostrictive alloys such as
Galfenol, steel additive |
| 65 |
Tb |
Terbium |
after the village of Ytterby, Sweden. |
Green
phosphors,
lasers,
fluorescent lamps,
magnetostrictive alloys such as
Terfenol-D |
| 66 |
Dy |
Dysprosium |
from the Greek "dysprositos", meaning hard to get. |
Rare-earth magnets,
lasers,
magnetostrictive alloys such as
Terfenol-D |
| 67 |
Ho |
Holmium |
after
Stockholm (in Latin, "Holmia"), native city of one of its
discoverers. |
Lasers, wavelength calibration standards for optical
spectrophotometers,
magnets |
| 68 |
Er |
Erbium |
after the village of Ytterby, Sweden. |
Infrared
lasers,
vanadium steel,
fiber-optic technology |
| 69 |
Tm |
Thulium |
after the mythological northern land of
Thule. |
Portable
X-ray machines,
metal-halide lamps,
lasers |
| 70 |
Yb |
Ytterbium |
after the village of Ytterby, Sweden. |
Infrared
lasers, chemical
reducing agent,
decoy flares,
stainless steel, stress gauges,
nuclear medicine |
| 71 |
Lu |
Lutetium |
after
Lutetia, the city which later became
Paris. |
Positron emission tomography - PET scan detectors, high
refractive index glass,
lutetium tantalate hosts for phosphors |
Abbreviations
The following abbreviations are often used:
- RE = rare earth
- REM = rare-earth metals
- REE = rare-earth elements
- REO = rare-earth oxides
- REY = rare-earth elements and yttrium
- LREE = light rare earth elements (La, Ce, Pr, Nd, Pm, Sm, Eu,
and Gd; also known as the cerium group)[6][7]
- HREE = heavy rare earth elements (Tb, Dy, Ho, Er, Tm, Yb, Lu,
and Y; also known as the yttrium group)[6][7]
Discovery
and early history
Rare earth elements became known to the world with the discovery of
the black mineral "ytterbite" (renamed to
gadolinite in 1800) by Lieutenant
Carl Axel Arrhenius in 1787, at a quarry in the village of
Ytterby, Sweden.[8]
Arrhenius' "ytterbite" reached
Johan Gadolin, a
Royal Academy of Turku professor, and his analysis yielded an
unknown oxide (earth) which he called
yttria.
Anders Gustav Ekeberg isolated
beryllium from the gadolinite but failed to recognize other elements
which the ore contained. After this discovery in 1794 a mineral from
Bastnäs
near
Riddarhyttan, Sweden, which was believed to be an
iron-tungsten
mineral, was re-examined by
Jöns Jacob Berzelius and
Wilhelm Hisinger. In 1803 they obtained a white oxide and called it
ceria.
Martin Heinrich Klaproth independently discovered the same oxide and
called it ochroia.
Thus by 1803 there were two known rare earth elements, yttrium
and cerium, although it took another 30 years for researchers to
determine that other elements were contained in the two ores ceria and
yttria (the similarity of the rare earth metals' chemical properties
made their separation difficult).
In 1839
Carl Gustav Mosander, an assistant of Berzelius, separated ceria by
heating the nitrate and dissolving the product in
nitric acid. He called the oxide of the soluble salt lanthana.
It took him three more years to separate the lanthana further into
didymia and pure lanthana. Didymia, although not further separable
by Mosander's techniques was a mixture of oxides.
In 1842 Mosander also separated the yttria into three oxides: pure
yttria, terbia and erbia (all the names are derived from the town name
"Ytterby"). The earth giving pink salts he called terbium; the
one which yielded yellow peroxide he called erbium.
So in 1842 the number of rare earth elements had reached six:
yttrium, cerium, lanthanum, didymium, erbium
and terbium.
Nils Johan Berlin and
Marc Delafontaine tried also to separate the crude yttria and found
the same substances that Mosander obtained, but Berlin named (1860) the
substance giving pink salts erbium and Delafontaine named the
substance with the yellow peroxide terbium. This confusion led to
several false claims of new elements, such as the mosandrium of
J. Lawrence Smith, or the philippium and decipium of
Delafontaine.
Spectroscopy
There were no further discoveries for 30 years, and the element
didymium was listed in the periodic table of elements with a
molecular mass of 138. In 1879 Delafontaine used the new physical
process of optical-flame
spectroscopy, and he found several new spectral lines in didymia.
Also in 1879, the new element
samarium was isolated by
Paul Émile Lecoq de Boisbaudran from the mineral
samarskite.
The samaria earth was further separated by Lecoq de Boisbaudran in
1886 and a similar result was obtained by
Jean Charles Galissard de Marignac by direct isolation from
samarskite. They named the element
gadolinium after
Johan Gadolin, and its oxide was named "gadolinia".
Further spectroscopic analysis between 1886 and 1901 of samaria,
yttria, and samarskite by
William Crookes, Lecoq de Boisbaudran and
Eugène-Anatole Demarçay yielded several new spectroscopic lines that
indicated the existence of an unknown element. The fractional
crystallization of the oxides then yielded
europium in 1901.
In 1839 the third source for rare earths became available. This is a
mineral similar to gadolinite, uranotantalum (now called
"samarskite"). This mineral from
Miass in
the southern
Ural Mountains was documented by Gustave Rose. The Russian chemist
R. Harmann proposed that a new element he called "ilmenium"
should be present in this mineral, but later,
Christian Wilhelm Blomstrand, Galissard de Marignac, and
Heinrich Rose found only
tantalum and
niobium
(columbium)
in it.
The exact number of rare earth elements that existed was highly
unclear, and a maximum number of 25 was estimated. The use of X-ray
spectra (obtained by
X-ray crystallography) by
Henry Gwyn Jeffreys Moseley made it possible to assign atomic
numbers to the elements. Moseley found that the exact number of
lanthanides had to be 15 and that
element 61 had yet to be discovered.
Using these facts about atomic numbers from X-ray crystallography,
Moseley also showed that
hafnium
(element 72) would not be a rare earth element. Moseley was killed in
World War I in 1915, years before hafnium was discovered. Hence, the
claim of
Georges Urbain that he had discovered element 72 was untrue. Hafnium
is an element that lies in the periodic table immediately below
zirconium, and hafnium and zirconium are very similar in their
chemical and physical properties.
During the 1940s,
Frank Spedding and others in the
United States (during the
Manhattan Project) developed the chemical
ion exchange procedures for separating and purifying the rare earth
elements. This method was first applied to the
actinides for separating
plutonium-239 and
neptunium, from
uranium,
thorium,
actinium, and the other actinide rare earths in the materials
produced in
nuclear reactors. The plutonium-239 was very desirable because it is
a
fissile material.
The principal sources of rare earth elements are the minerals
bastnäsite,
monazite, and
loparite and the
lateritic ion-adsorption
clays.
Despite their high relative abundance,
rare earth minerals are more difficult to mine and extract than
equivalent sources of
transition metals (due in part to their similar chemical
properties), making the rare earth elements relatively expensive. Their
industrial use was very limited until efficient separation techniques
were developed, such as
ion exchange,
fractional crystallization and
liquid-liquid extraction during the late 1950s and early 1960s.[9]
Early
classification
Before the time that
ion exchange methods and
elution
were available, the separation of the rare earths was primarily achieved
by repeated
precipitation or
crystallisation. In those days, the first separation was into two
main groups, the cerium group earths (scandium, lanthanum, cerium,
praseodymium, neodymium, and samarium) and the yttrium group earths
(yttrium, dysprosium, holmium, erbium, thulium, ytterbium, and
lutetium). Europium, gadolinium, and terbium were either considered as a
separate group of rare earth elements (the terbium group), or europium
was included in the cerium group, and gadoliniun and terbium were
included in the yttrium group. The reason for this division arose from
the difference in
solubility of rare earth double sulfates with sodium and potassium.
The sodium double sulfates of the cerium group are difficultly soluble,
those of the terbium group slightly, and those of the yttrium group are
very soluble.[10]
Origin
Rare earth elements are heavier than
iron and
thus are produced by
supernova nucleosynthesis or the
s-process in
asymptotic giant branch stars. In nature,
spontaneous fission of
uranium-238 produces trace amounts of radioactive
promethium, but most promethium is synthetically produced in nuclear
reactors.
Rare earth elements change through time in small quantities (ppm,
parts per million), so their proportion can be used for
geochronology and dating fossils.
Geological
distribution
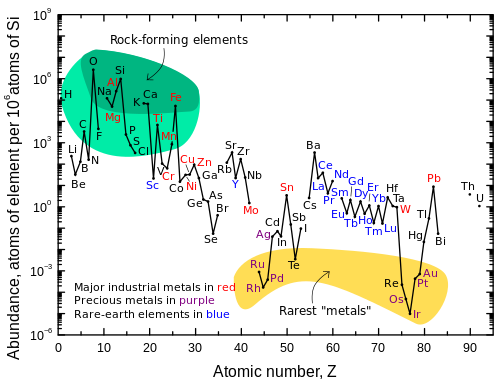
Abundance of elements in the Earth crust per million of Si
atoms
Rare earth
cerium
is actually the 25th most abundant element in the
Earth's crust, having 68 parts per million (about as common as
copper). Only the highly unstable and radioactive
promethium "rare earth" is quite scarce.
The rare earth elements are often found together. The longest-lived
isotope of promethium has a half life of 17.7 years, so the element
exists in nature in only negligible amounts (approximately 572 g in the
entire Earth's crust).[11]
Promethium is one of the two elements that do not have stable
(non-radioactive) isotopes and are followed by (i.e. with higher atomic
number) stable elements.
Due to
lanthanide contraction, yttrium, which is trivalent, is of similar
ionic size to
dysprosium and its lanthanide neighbors. Due to the relatively
gradual decrease in ionic size with increasing atomic number, the rare
earth elements have always been difficult to separate. Even with eons of
geological time, geochemical separation of the lanthanides has only
rarely progressed much farther than a broad separation between light
versus heavy lanthanides, otherwise known as the cerium and yttrium
earths. This geochemical divide is reflected in the first two rare
earths that were discovered,
yttria in 1794 and
ceria in 1803. As originally found, each comprised the entire
mixture of the associated earths. Rare earth minerals, as found, usually
are dominated by one group or the other, depending upon which size-range
best fits the structural lattice. Thus, among the anhydrous rare earth
phosphates, it is the tetragonal mineral
xenotime that incorporates yttrium and the yttrium earths, whereas
the monoclinic
monazite phase incorporates cerium and the cerium earths
preferentially. The smaller size of the yttrium group allows it a
greater solid solubility in the rock-forming minerals that comprise the
Earth's mantle, and thus yttrium and the yttrium earths show less
enrichment in the Earth's crust relative to chondritic abundance, than
does cerium and the cerium earths. This has economic consequences: large
ore bodies of the cerium earths are known around the world, and are
being exploited. Corresponding orebodies for yttrium tend to be rarer,
smaller, and less concentrated. Most of the current supply of yttrium
originates in the "ion absorption clay" ores of Southern China. Some
versions provide concentrates containing about 65% yttrium oxide, with
the heavy lanthanides being present in ratios reflecting the
Oddo-Harkins rule: even-numbered heavy lanthanides at abundances of
about 5% each, and odd-numbered lanthanides at abundances of about 1%
each. Similar compositions are found in xenotime or gadolinite.
Well-known minerals containing yttrium include gadolinite, xenotime,
samarskite,
euxenite,
fergusonite, yttrotantalite, yttrotungstite, yttrofluorite (a
variety of
fluorite), thalenite,
yttrialite. Small amounts occur in
zircon,
which derives its typical yellow fluorescence from some of the
accompanying heavy lanthanides. The
zirconium mineral
eudialyte, such as is found in southern
Greenland, contains small but potentially useful amounts of yttrium.
Of the above yttrium minerals, most played a part in providing research
quantities of lanthanides during the discovery days.
Xenotime is occasionally recovered as a byproduct of heavy sand
processing, but is not as abundant as the similarly recovered monazite
(which typically contains a few percent of yttrium). Uranium ores from
Ontario have occasionally yielded yttrium as a byproduct.
Well-known minerals containing cerium and the light lanthanides
include
bastnäsite,
monazite,
allanite,
loparite,
ancylite,
parisite,
lanthanite, chevkinite,
cerite,
stillwellite, britholite,
fluocerite, and cerianite. Monazite (marine sands from
Brazil,
India, or
Australia; rock from
South Africa), bastnäsite (from
Mountain Pass, California, or several localities in China), and
loparite (Kola
Peninsula,
Russia)
have been the principal ores of cerium and the light lanthanides.
In 2011, Yasuhiro Kato, a geologist at the
University of Tokyo who led a study of Pacific Ocean seabed mud,
published results indicating the mud could hold rich concentrations of
rare earth minerals. The deposits, studied at 78 sites, came from "[h]ot
plumes from hydrothermal vents pull[ing] these materials out of seawater
and deposit[ing] them on the seafloor, bit by bit, over tens of millions
of years. One square patch of metal-rich mud 2.3 kilometers wide might
contain enough rare earths to meet most of the global demand for a year,
Japanese geologists report July 3 in
Nature Geoscience." "I believe that rare earth resources
undersea are much more promising than on-land resources," said Kato.
"[C]oncentrations of rare earths were comparable to those found in clays
mined in China. Some deposits contained twice as much heavy rare earths
such as dysprosium, a component of magnets in hybrid car motors."[12]
Global
rare earth production

Global production 1950–2000
Until 1948, most of the world's rare earths were sourced from
placer sand deposits in
India and
Brazil.[13]
Through the 1950s, South Africa took the status as the world's rare
earth source, after large veins of rare earth bearing
monazite were discovered there.[13]
Through the 1960s until the 1980s, the
Mountain Pass rare earth mine in California was the leading
producer. Today, the Indian and South African deposits still produce
some rare earth concentrates, but they are dwarfed by the scale of
Chinese production. China had produced over 95% of the world's rare
earth supply, mostly in
Inner Mongolia,[3][14]
even though it had only 37% of
proven reserves,[15]
although these numbers have since been reported to have slipped to 90%
and 23%, respectively, by 2012.[16]
All of the world's heavy rare earths (such as dysprosium) come from
Chinese rare earth sources such as the
polymetallic
Bayan Obo deposit.[14][17]
In 2010, the
United States Geological Survey (USGS) released a study which found
that the United States had 13 million metric tons of rare earth
elements.[18]
New demand has recently strained supply, and there is growing concern
that the world may soon face a shortage of the rare earths.[19]
In several years from 2009 worldwide demand for rare earth elements is
expected to exceed supply by 40,000 tonnes annually unless major new
sources are developed.[20]
China
These concerns have intensified due to the actions of China, the
predominant supplier.[21]
Specifically, China has announced regulations on exports and a crackdown
on smuggling.[22]
On September 1, 2009, China announced plans to reduce its export quota
to 35,000 tons per year in 2010–2015, ostensibly to conserve scarce
resources and protect the environment.[23]
On October 19, 2010
China Daily, citing an unnamed Ministry of Commerce official,
reported that China will "further reduce quotas for rare earth exports
by 30 percent at most next year to protect the precious metals from
over-exploitation".[24]
At the end of 2010 China announced that the first round of export quotas
in 2011 for rare earths would be 14,446 tons which was a 35% decrease
from the previous first round of quotas in 2010.[25]
China announced further export quotas on 14 July 2011 for the second
half of the year with total allocation at 30,184 tons with total
production capped at 93,800 tonnes.[26]
In September 2011 China announced the halt in production of three of its
eight major rare earth mines, responsible for almost 40% of China's
total rare earth production.
In March 2012, the U.S., E.U., and Japan confronted China at WTO about
these export and production restrictions. China responded with claims
that the restrictions had environmental protection in mind.[28]
In August 2012, China announced a further 20% reduction in production.[29]
These restrictions have damaged industries in other countries and forced
producers of rare earth products to relocate their operations to China.[28]
The Chinese restrictions on supply failed in 2012 as prices dropped in
response to the opening of other sources.[30]
Outside of China
As a result of the increased demand and tightening restrictions on
exports of the metals from China, some countries are stockpiling rare
earth resources.[31]
Searches for alternative sources in
Australia,
Brazil,
Canada,
South Africa,
Tanzania,
Greenland, and the
United States are ongoing.[32]
Mines in these countries were closed when China undercut world prices in
the 1990s, and it will take a few years to restart production as there
are many
barriers to entry.[22]
One example is the
Mountain Pass mine in
California, that announced its resumption of operations on a
start-up basis on August 27, 2012.[14][33]
Other significant sites under development outside of China include the
Nolans Project in Central Australia, the remote
Hoidas Lake project in northern Canada,[34]
and the
Mount Weld project in Australia.[14][33][35]
The
Hoidas Lake project has the potential to supply about 10% of the $1
billion of REE consumption that occurs in North America every year.[36]
Vietnam
signed an agreement in October 2010 to supply Japan with rare earths[37]
from its
northwestern
Lai Châu Province.[38]
Also under consideration for mining are sites such as
Thor
Lake in the
Northwest Territories, various locations in
Vietnam,[14][20]
and a site in southeast
Nebraska in the US, where Quantum Rare Earth Development, a Canadian
company, is currently conducting test drilling and economic feasibility
studies toward opening a niobium mine.[39]
Additionally, a large deposit of rare earth minerals was recently
discovered in
Kvanefjeld in southern
Greenland.[40]
Pre-feasibility drilling at this site has confirmed significant
quantities of black
lujavrite, which contains about 1% rare earth oxides (REO).[41]
The
European Union has urged Greenland to restrict Chinese development
of rare-earth projects there, but as of early 2013, the
government of Greenland has said that it has no plans to impose such
restrictions.[42]
Many Danish politicians have expressed concerns that other nations,
including China, could gain influence in thinly populated Greenland,
given the number of foreign workers and investment that could come from
Chinese companies in the near future because of the law passed December
2012.[43]
Adding to potential mine sites,
ASX listed Peak Resources announced in February 2012, that their
Tanzanian based
Ngualla
project contained not only the 6th largest deposit by tonnage outside of
China, but also the highest grade of rare earth elements of the 6.[44]
Other sources
Significant quantities of rare earth oxides are found in tailings
accumulated from 50 years of
uranium ore,
shale and
loparite mining at
Sillamäe,
Estonia.[45]
Due to the rising prices of rare earths, extraction of these oxides has
become economically viable. The country currently exports around 3,000
tonnes per year, representing around 2% of world production.[46]
Similar resources are suspected in the western United States, where
gold
rush-era mines are believed to have discarded large amounts of rare
earths, since they had no value at the time.[47]
Nuclear reprocessing is another potential source of rare earth or
any other elements.
Nuclear fission of
uranium
or
plutonium produces a full range of elements, including all their
isotopes. However, due to the radioactivity of many of these
isotopes, it is unlikely that extracting them from the mixture can be
done safely and economically.
In May 2012, researchers from two prevalent universities in Japan
announced that they had discovered rare earths in
Ehime Prefecture, Japan.[48][49]
In 2012, Japanese scientists discovered about 6.8 million tons of rare
earth elements near the island of
Minami-Tori-Shima, enough to supply Japan's current consumption for
over 200 years. Around 90% of the world's production of REE comes from
China, and Japan imports 60% of that.[50]
Recycling
Another recently developed source of rare earths is
electronic waste and other
wastes
that have significant rare earth components. New advances in
recycling technology have made extraction of rare earths from these
materials more feasible,[51]
and recycling plants are currently operating in Japan, where there is an
estimated 300,000 tons of rare earths stored in unused electronics.[52]
In France,
the
Rhodia group is setting up two factories, in
La Rochelle and
Saint-Fons, that will produce 200 tons a year of rare earths from
used
fluorescent lamps, magnets and batteries.[53][54]
Refining
In early 2011, Australian mining company,
Lynas,
was reported to be "hurrying to finish" a US$230 million rare earth
refinery on the eastern coast of Malaysia's industrial port of
Kuantan.
The plant would refine ore— Lanthanide concentrate from the
Mount Weld mine in Australia. The ore would be trucked to
Fremantle and transported by
container ship to Kuantan. However, the Malaysian authorities
confirmed that as of October 2011, Lynas was not given any permit to
import any rare earth ore into Malaysia. On February 2, 2012, the
Malaysian AELB (Atomic Energy Licensing Board) recommended that Lynas be
issued a Temporary Operating License (TOL) subject to completion of a
number of conditions. On April 3, 2012, Lynas announced to the Malaysian
media that these conditions had been met, and was now waiting on the
issuance of the licence. Within two years, Lynas was said to expect the
refinery to be able to meet nearly a third of the world's demand for
rare earth materials, not counting
China."[55]
The Kuantan development brought renewed attention to the Malaysian town
of
Bukit Merah in
Perak,
where a rare-earth mine operated by a
Mitsubishi Chemical subsidiary, Asian Rare Earth, closed in 1992 and
left
continuing environmental and health concerns.[56]
In mid-2011, after
protests, Malaysian government restrictions on the Lynas plant were
announced. At that time, citing subscription-only
Dow Jones Newswire reports, a
Barrons report said the Lynas investment was $730 million, and
the projected share of the global market it would fill put at "about a
sixth."[57]
An independent review was initiated by Malaysian Government and
United Nations and conducted by the
International Atomic Energy Agency (IAEA) between 29 May and 3 June
2011 to address concerns of radioactive hazards. The IAEA team was not
able to identify any non-compliance with international radiation safety
standards.[58]
Environmental considerations
Mining, refining, and recycling of rare earths have serious
environmental consequences if not properly managed. A particular hazard
is mildly radioactive slurry
tailings resulting from the common occurrence of
thorium
and
uranium in rare earth element ores.[59]
Additionally, toxic acids are required during the refining process.[15]
Improper handling of these substances can result in extensive
environmental damage. In May 2010, China announced a major, five-month
crackdown on illegal mining in order to protect the environment and its
resources. This campaign is expected to be concentrated in the South,[60]
where mines – commonly small, rural, and illegal operations – are
particularly prone to releasing toxic wastes into the general water
supply.[14][61]
However, even the major operation in
Baotou,
in Inner Mongolia, where much of the world's rare earth supply is
refined, has caused major environmental damage.[15]
The
Bukit Merah mine in Malaysia has been the focus of a US$100 million
cleanup which is proceeding in 2011. "Residents blamed a rare earth
refinery for
birth defects and eight
leukemia cases within five years in a community of 11,000 — after
many years with no leukemia cases." Seven of the leukemia victims died.
After having accomplished the hilltop entombment of 11,000 truckloads of
radioactively contaminated material, the project is expected to entail
in summer, 2011, the removal of "more than 80,000 steel barrels of
radioactive waste to the hilltop repository." One of Mitsubishi's
contractors for the cleanup is
GeoSyntec, an
Atlanta-based
firm.[56]
Osamu Shimizu, a director of Asian Rare Earth, said, "the company might
have sold a few bags of calcium phosphate fertilizer on a trial basis as
it sought to market byproducts," in reply to a former resident of Bukit
Merah who said, "The cows that ate the grass [grown with the fertilizer]
all died."[62]
In May 2011, after the
Fukushima Daiichi nuclear disaster, widespread protests took place
in Kuantan over the
Lynas refinery and radioactive waste from it. The ore to be
processed has very low levels of thorium, and Lynas founder and chief
executive Nicholas Curtis said "There is absolutely no risk to public
health." T. Jayabalan, a doctor who says he has been monitoring and
treating patients affected by the Mitsubishi plant, "is wary of Lynas's
assurances. The argument that low levels of thorium in the ore make it
safer doesn't make sense, he says, because radiation exposure is
cumulative."[62]
Construction of the facility has been halted until an independent
United Nations
IAEA panel investigation is completed, which is expected by the end
of June 2011.[63]
New restrictions were announced by the Malaysian government in late
June.[57]
IAEA panel investigation is completed and no construction has been
halted. Lynas is on budget and on schedule to start producing 2011. The
IAEA report has concluded in a report issued on Thursday June 2011 said
it did not find any instance of "any non-compliance with international
radiation safety standards" in the project.[64]
Geo-political considerations
China has officially cited resource depletion and environmental
concerns as the reasons for a nationwide crackdown on its rare earth
mineral production sector.
However, non-environmental motives have also been imputed to China's
rare earth policy.[15]
According to
The Economist, "Slashing their exports of rare-earth metals...is
all about moving Chinese manufacturers up the supply chain, so they can
sell valuable finished goods to the world rather than lowly raw
materials."[65]
One possible example is the division of General Motors which deals with
miniaturized magnet research, which shut down its US office and moved
its entire staff to
China in
2006
[66]
(it should be noted that China's export quota only applies to the metal
but not products made from these metals such as magnets).
It was reported,[67]
but officially denied,[68]
that China instituted an
export ban on shipments of rare earth oxides (but not alloys) to
Japan on 22 September 2010, in response to
the detainment of a Chinese fishing boat captain by the
Japanese Coast Guard.[69]
On September 2, 2010, a few days before the fishing boat incident,
The Economist reported that "China...in July announced the latest in
a series of annual export reductions, this time by 40% to precisely
30,258 tonnes."[70]
The
United States Department of Energy in its 2010 Critical Materials
Strategy report identified
dysprosium as the element that was most critical in terms of import
reliance.[71]
A 2011 report issued by the U.S. Geological Survey and U.S.
Department of the Interior, "China's Rare-Earth Industry," outlines
industry trends within China and examines national policies that may
guide the future of the country's production. The report notes that
China's lead in the production of rare-earth minerals has accelerated
over the past two decades. In 1990, China accounted for only 27% of such
minerals. In 2009, world production was 132,000 metric tons; China
produced 129,000 of those tons. According to the report, recent patterns
suggest that China will slow the export of such materials to the world:
"Owing to the increase in domestic demand, the Government has gradually
reduced the export quota during the past several years." In 2006, China
allowed 47 domestic rare-earth producers and traders and 12 Sino-foreign
rare-earth producers to export. Controls have since tightened annually;
by 2011, only 22 domestic rare-earth producers and traders and 9
Sino-foreign rare-earth producers were authorized. The government's
future policies will likely keep in place strict controls: "According to
China's draft rare-earth development plan, annual rare-earth production
may be limited to between 130,000 and 140,000 [metric tons] during the
period from 2009 to 2015. The export quota for rare-earth products may
be about 35,000 [metric tons] and the Government may allow 20 domestic
rare-earth producers and traders to export rare earths."[72]
The United States Geological Survey is actively surveying southern
Afghanistan for rare earth deposits under the protection of United
States military forces. Since 2009 the USGS has conducted remote sensing
surveys as well as fieldwork to verify Soviet claims that volcanic rocks
containing rare earth metals exist in Helmand province near the village
of Khanneshin. The USGS study team has located a sizable area of rocks
in the center of an extinct volcano containing light rare earth elements
including cerium and neodymium. It has mapped 1.3 million metric tons of
desirable rock, or about 10 years of supply at current demand levels.
The Pentagon has estimated its value at about $7.4 billion.[73]
Rare earth pricing
Rare earth elements are not exchange-traded in the same way that
precious (for instance,
gold and
silver)
or non-ferrous metals (such as
nickel,
tin,
copper,
and
aluminium) are. Instead they are sold on the private market, which
makes their prices difficult to monitor and track. The 17 elements are
not usually sold in their pure form, but instead are distributed in
mixtures of varying purity, e.g. "Neodymium metal ≥ 99%". As such,
pricing can vary based on the quantity and quality required by the end
user's application.
See also
References
-
^
"News and events". US Department of Agriculture.
Retrieved 2012-03-13.
-
^
Edited
by N G Connelly and T Damhus (with R M Hartshorn and A T
Hutton), ed. (2005).
Nomenclature of Inorganic Chemistry: IUPAC Recommendations
2005. Cambridge: RSC Publ.
ISBN 0-85404-438-8.
Archived from
the original on 2008-05-27.
Retrieved 2012-03-13.
- ^
a
b
"Haxel G, Hedrick J, Orris J. 2006. Rare earth elements critical
resources for high technology. Reston (VA): United States
Geological Survey. USGS Fact Sheet: 087‐02.".
Retrieved 2012-03-13.
-
^
a
b
c
C. R. Hammond, "Section 4; The
Elements", in CRC Handbook of Chemistry and Physics, 89th
Edition (Internet Version 2009), David R. Lide, ed., CRC
Press/Taylor and Francis, Boca Raton, FL.
-
^
Energy-efficient light bulbs containing yttrium
- ^
a
b
Gschneidner, Karl A., Jr. 1966.
|title=Rare Earths-The Fraternal Fifteen. Washington, DC, U.S.
atomic Energy Commission, Divisions of Technical Information, 42
pages.
- ^
a
b
Hedrick,
James B.
"REE Handbook -- The ultimate guide to Rare Earth Elements,".
Rare Metal Blog. Toronto, Canada.
-
^
Gschneidner KA, Cappellen, ed. (1987). "1787–1987 Two hundred
Years of Rare Earths". Rare Earth Information Center, IPRT,
North-Holland. IS-RIC 10.
-
^
Spedding F, Daane AH: "The Rare
Earths", John Wiley & Sons, Inc., 1961
-
^
B. Smith Hopkins: "Chemistry of the
rarer elements", D. C. Heath & Company, 1923
-
^
P.
Belli, R. Bernabei, F. Cappella, R. Cerulli, C.J. Dai, F.A.
Danevich, A. d'Angelo, A. Incicchitti, V.V. Kobychev, S.S.
Nagorny, S. Nisi, F. Nozzoli, D. Prosperi, V.I. Tretyak, S.S.
Yurchenko (2007). "Search for α decay of natural Europium".
Nuclear Physics A 789 (1–4): 15–29.
Bibcode:2007NuPhA.789...15B.
doi:10.1016/j.nuclphysa.2007.03.001.
-
^
Powell, Devin,
"Rare earth elements plentiful in ocean sediments",
ScienceNews, July 3rd, 2011. Via Kurt Brouwer's
Fundmastery Blog, MarketWatch, 2011-07-05.. Retrieved
2011-07-05.
- ^
a
b
ER, Rose. Rare Earths of the
Grenville Sub‐Province Ontatio and Quebec. GSC Report Number
59‐10. Ottawa: Geological Survey of Canada Department of Mines
and Technical Surveys, 1960.
- ^
a
b
c
d
e
f
China's Rare Earth Dominance, Wikinvest. Retrieved on 11 Aug
2010.
- ^
a
b
c
d
Bradsher, Keith (October 29, 2010).
"After China's Rare Earth Embargo, a New Calculus". The
New York Times. Retrieved
October 30, 2010.
-
^
China Warns its Rare Earth Reserves are Declining BBC News
June 20, 2012. Retrieved June 20, 2012
-
^
Chao ECT, Back JM, Minkin J,
Tatsumoto M, Junwen W, Conrad JE, McKee EH, Zonglin H, Qingrun
M.
"Sedimentary carbonate‐hosted giant Bayan Obo REE‐Fe‐Nb ore
deposit of Inner Mongolia, China; a cornerstone example for
giant polymetallic ore deposits of hydrothermal origin."
1997. United States Geological Survey Publications Warehouse. 29
February 2008.
-
^
USGS.
Rare Earth Elements in U.S. Not So Rare: Significant Deposits
Found in 14 States. U.S. Department of the Interior. Full
Report:
The Principal Rare Earth Elements Deposits of the United
States—A Summary of Domestic Deposits and a Global Perspective.
-
^
"Cox C. 2008. Rare earth innovation. Herndon (VA): The Anchor
House Inc;". Retrieved
2008-04-19.
- ^
a
b
"As hybrid cars gobble rare metals, shortage looms".
Reuters. August 31, 2009. Retrieved Aug 31, 2009
-
^
How Beijing Cornered the Rare Earths Market April 25, 2012
Foreign Affairs
-
^
a
b
Livergood R. (2010).
Rare Earth Elements: A Wrench in the Supply Chain.
Center for Strategic and International Studies.
Retrieved 2012-03-13.
-
^
China To Limit Rare Earths Exports, Manufacturing.net, 1
September 2009. Retrieved 2010-08-30.
-
^
China to cut exports of rare earth minerals vital to energy
tech" thehill.com, 19 Oct. 2009. Retrieved 2010-10-19.
-
^
China's Rare Earth Exports Surge in Value"
thechinaperspective.com, January 19. 2011
-
^
Zhang,
Ding, Fu, Qi, Qingfen, Jing.
"Rare earths export quota unchanged". ChinaDaily.com.cn.
Retrieved 2011-07-15.
- ^
a
b
"WRAPUP 4-US, EU, Japan take on China at WTO over rare earths" -
Reuters, March 13, 2012
-
^
CNN China cuts mines vital to tech industry
-
^
"El Reg man: Too bad, China - I was RIGHT about hoarding rare
earths."
-
^
EU stockpiles rare earths as tensions with china rise,
Financial Post, retrieved
2011-09-07
-
^
"Canadian Firms Step Up Search for Rare-Earth Metals".
NYTimes.com (Reuters). 2009-09-09.
Retrieved 2009-09-15.
-
^
a
b
Leifert, H. Restarting U.S. rare
earth production?. Earth magazine. June 2010. Pgs 20–21.
-
^
"Lunn J. 2006. Great western minerals. London: Insigner Beaufort
Equity Research".
Retrieved 2008-04-19.
-
^
Gorman,
Steve (2009-08-31).
"California mine digs in for 'green' gold rush.". Reuters.
Retrieved 2010-03-22.
-
^
"Hoidas Lake Project".
Retrieved 2008-09-24.
-
^
"Rare earths supply deal between Japan and Vietnam". BBC
News. 31 October 2010.
-
^
"Vietnam signs major nuclear pacts".
AlJazeera. 31 October 2010.
Retrieved 31 October 2010.
-
^
"High-tech buried treasure.".
Retrieved 2010-05-05.
-
^
Greenland
"Rare Earth Elements at Kvanefjeld, Greenland", Retrieved on
2010-11-10.
-
^
Greenland
"New Multi-Element Targets and Overall Resource Potential",
Retrieved on 2010-11-10.
-
^
Chinese Workers—in Greenland? February 10, 2013
BusinessWeek
-
^
Greenland Votes to Get Tough on Investors; New Ruling Party
Campaigned to Backtrack on Country's Recent Opening to
Investment From Foreign Mining Ventures March 13, 2013
WSJ
-
^
Peak Resources – Maiden Resource, Ngualla Rare Earth Project,
ASX Announcement, 29th February 2012
-
^
Rofer,
Cheryl K.; Tõnis Kaasik (2000). Turning a Problem Into a
Resource: Remediation and Waste Management at the Sillamäe Site,
Estonia. Volume 28 of NATO science series: Disarmament
technologies. Springer. p. 229.
ISBN 978-0-7923-6187-9.
-
^
Anneli
Reigas (2010-11-30).
"Estonia's rare earth break China's market grip". AFP.
Retrieved 2010-12-01.
-
^
Cone,
Tracie (July 21, 2013).
"Gold Rush Trash is Information Age Treasure". USA Today.
Retrieved July 21, 2013.
-
^
"Japan Discovers Domestic Rare Earths Reserve". BrightWire.
-
^
http://www.brightwire.com/news/search?utf8=%E2%9C%93&fuzzy_path=%2Ffuzzy_search%2Fentity&q=rare+earth+
-
^ Westlake, Adam.
"Scientists in Japan discover rare earths in Pacific Ocean east
of Tokyo"
Japan Daily Press, 29 June 2012. Retrieved: 29 June
2012.
-
^
Recycling of neodymium and samarium
-
^
Tabuchi, Hiroko. "Japan
Recycles Minerals From Used Electronics".
New York Times. October 5, 2010.
-
^
Rhodia press release
http://www.rhodia.com/en/news_center/news_releases/Recycle_rare_earths_031011.tcm
-
^
[1]
-
^
Bradsher, Keith,
"Taking a Risk for Rare Earths", The New York Times,
March 8, 2011 (March 9, 2011 p. B1 NY ed.). Retrieved
2011-03-09.
- ^
a
b
Bradsher, Keith,
"Mitsubishi Quietly Cleans Up Its Former Refinery", The
New York Times, March 8, 2011 (March 9, 2011 p. B4 NY ed.).
Retrieved 2011-03-09.
- ^
a
b
Coleman, Murray,"Rare
Earth ETF Jumps As Plans To Break China's Hold Suffer Setback",
Barrons blog, June 30, 2011 1:52 PM ET. Retrieved
2011=-6-30.
-
^
Report of the International Review Mission on the Radiation
Safety Aspects of a Proposed Rare Earths Processing Facility
(Lynas Project). (PDF) . Retrieved on 2011-09-27.
-
^
Bourzac, Katherine.
"Can the U.S. Rare-Earth Industry Rebound?" Technology
Review. October 29, 2010.
-
^
Govt cracks whip on rare earth mining. China Daily,
May 21, 2010. Accessed June 3rd, 2010.
-
^
Y, Lee. "South
China Villagers Slam Pollution From Rare Earth Mine." 22
February 2008. RFA English Website. 16 March 2008
- ^
a
b
Lee, Yoolim,
"Malaysia Rare Earths in Largest Would-Be Refinery Incite
Protest",
Bloomberg Markets Magazine, May 31, 2011 5:00 PM ET.
-
^
"UN investigation into Malaysia rare-earth plant safety",
BBC,
30 May 2011 05:52 ET.
-
^
IAEA Submits Lynas Report to Malaysian Government. Iaea.org
(2011-06-29). Retrieved on 2011-09-27.
-
^
"The Difference Engine: More precious than gold".
The Economist September 17, 2010.
-
^
C, Cox.
"Rare earth innovation: the silent shift to China". 16
November 2006. The Anchor House: Research on Rare Earth Elements
Accessed 29 February 2008
-
^
Bradsher, Keith (2010-09-22).
"Amid Tension, China Blocks Vital Exports to Japan". The New
York Times Company. Retrieved
22 September 2010.
-
^
James T. Areddy, David Fickling And
Norihiko Shirouzu (2010-09-23).
"China Denies Halting Rare-Earth Exports to Japan". Wall
Street Journal. Retrieved 22
September 2010.
-
^
Backlash over the alleged China curb on metal exports,
Daily Telegraph, London, 29 Aug 2010. Retrieved
2010-08-30.
-
^
"Rare earths: Digging in"
The Economist September 2, 2010
-
^
Mills, Mark P.
"Tech's Mineral Infrastructure – Time to Emulate China's Rare
Earth Policies."
Forbes, 1 January 2010.
-
^
"U.S. Geological Survey: China's Rare-Earth Industry".
Journalist's Resource.org.
-
^
Simpson, S.: Afghanistan's Buried
Riches, "Scientific American", October 2011
External links
- Tabuchi, Hiroko (5 October 2010).
"Japan Recycles Rare Earth Minerals From Used Electronics".
The New York Times.
- Kan, Michael (7 October 2010).
"Common gadgets may be affected by shortage of rare earths". New
Zealand PC World Magazine.
Retrieved 6 October 2010.
- Auslin, Michael (13 October 2010).
"Japan's Rare-Earth Jolt". Wall Street Journal.
Retrieved 13 October 2010.
- Aston, Adam (15 October 2010).
"China's Rare-Earth Monopoly". Technology Review (MIT).
Retrieved 17 October 2010.
- Hurst, Cindy (March 2010).
"China's Rare Earth Elements Industry: What Can the West Learn?".
Institute for the Analysis of Global Security (IAGS).
Retrieved 18 October 2010.
-
Rare earths mining: China's 21st Century gold rush, BBC News
June 2010 infographic examining China's role in the rare earths
market.
-
Rare Earth Elements in National Defense: Background, Oversight
Issues, and Options for Congress Congressional Research Service,
March 31, 2011.
-
Digging for rare earths: The mines where iPhones are born | Apple -
CNET News, September 26, 2012
- Khan, Malek; Lundmark, Martin; Hellström, Jerker
"Rare Earth Elements and Europe’s Dependence on China" in
Strategic Outlook 2013, Swedish Defence Research Agency (FOI),
June 2013, pp.93-98.

|
|
|
DA INGLESE A ITALIANO
Inserire
nella casella Traduci la parola
INGLESE e cliccare
Go.
DA ITALIANO A INGLESE
Impostare INGLESE anziché italiano e
ripetere la procedura descritta.
|
|